Testsealabs COVID-19 ANTIGEN TEST CASSETTE(SWAB)
【INTENDED USE】
Testsealabs®COVID-19 Antigen Test Cassette is a rapid chromatographic immunoassay for the qualitative detection of COVID-19 antigen in nasal swab specimen to aid in the diagnosis of COVID-19 viral infection.
【Specification】
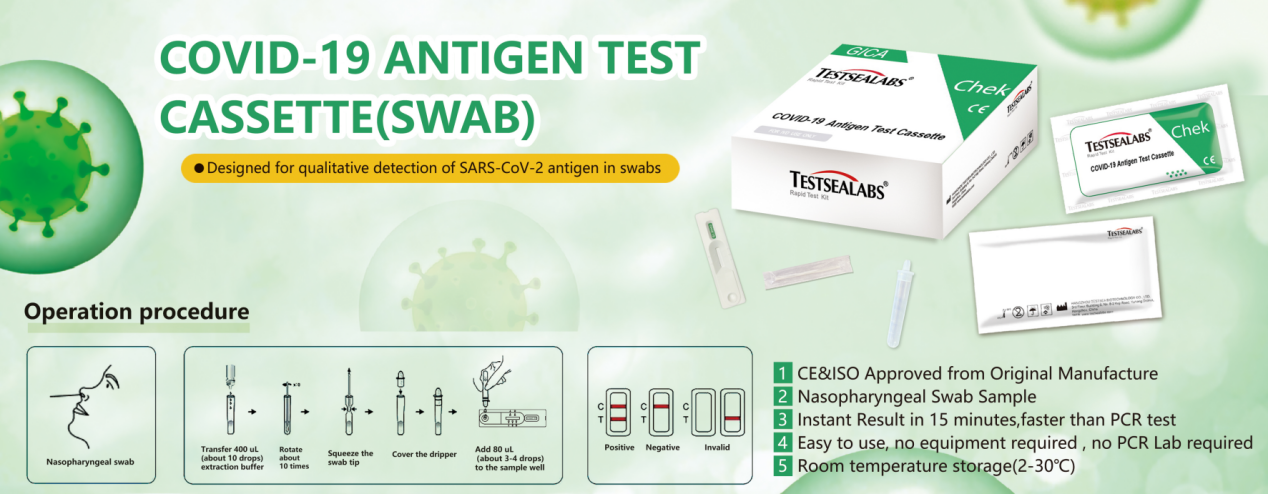
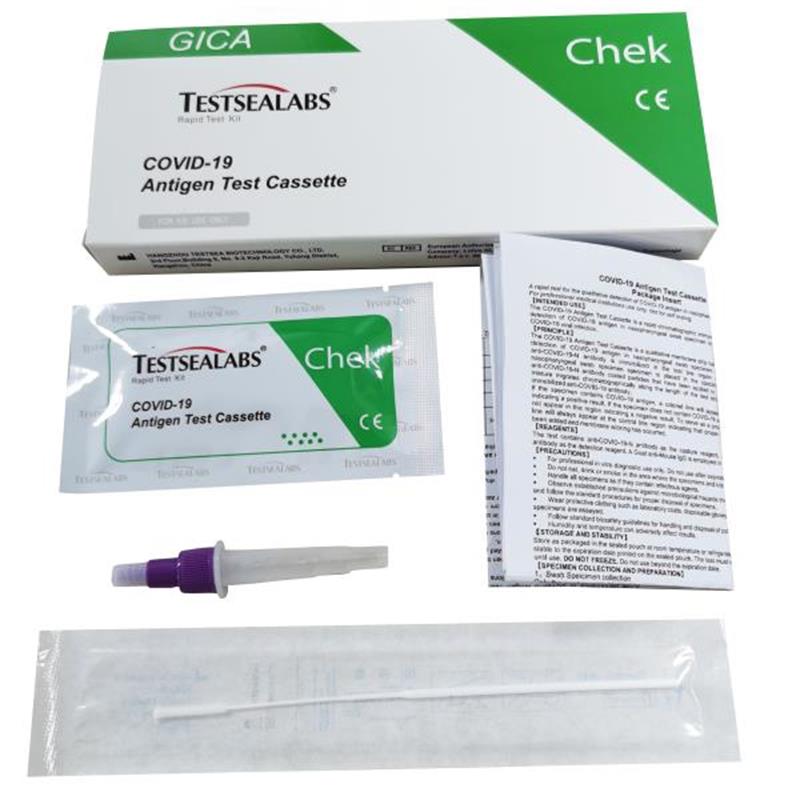
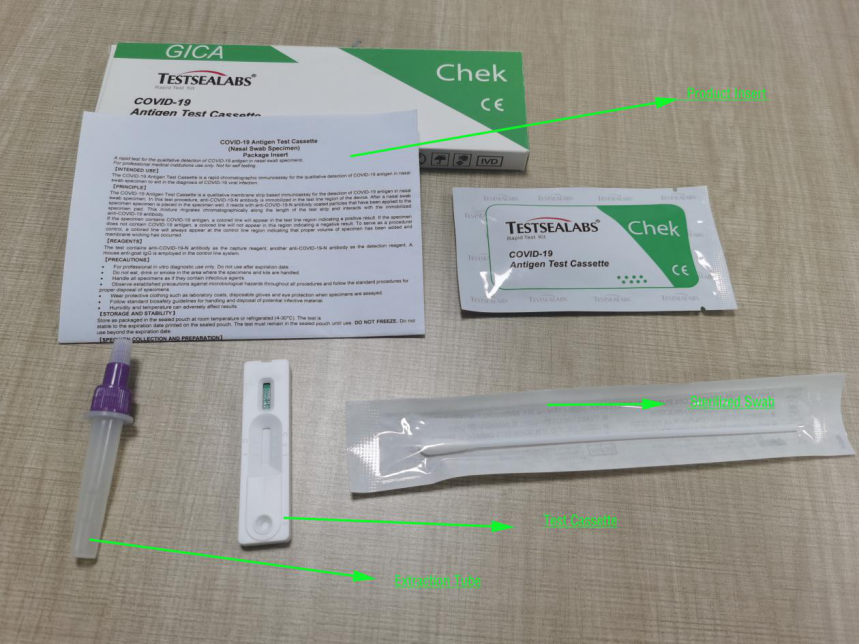
1pc/box ( 1 test device+ 1 Sterilized Swab+1 Extraction Buffer+1 Product Insert)
【MATERIALS PROVIDED】
1.Test Devices
2.Extraction Buffer
3.Sterilized Swab
4.Package Insert
【SPECIMENS COLLECTION】
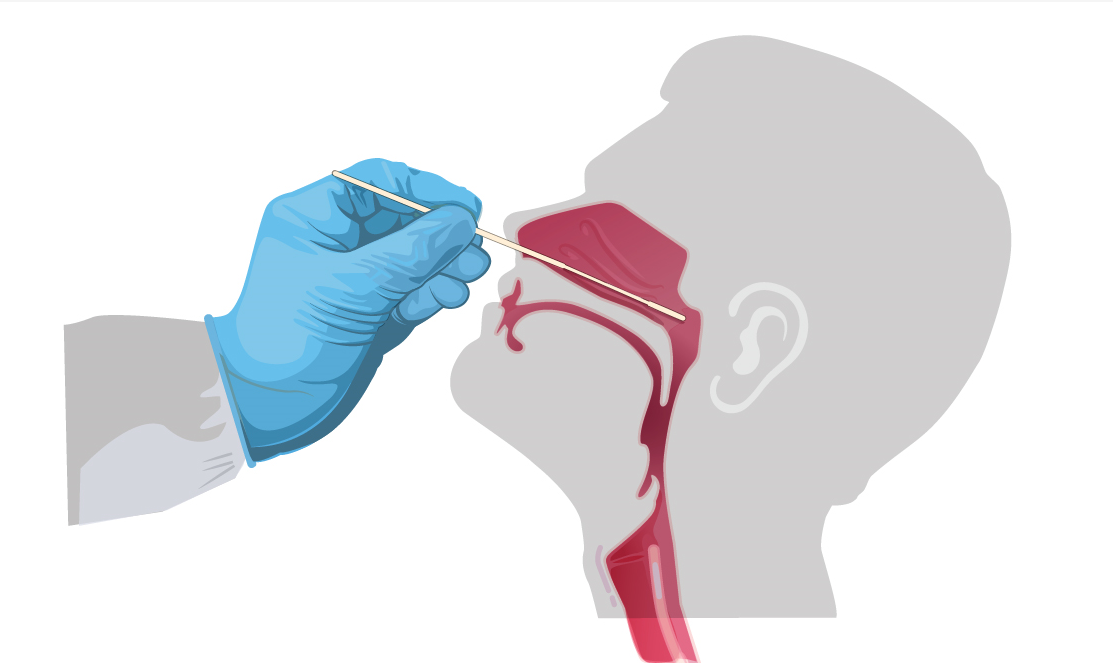
Insert mini tip swab with a flexible shaft (wire or plastic) through the nostril parallel to the palate (not upwards) until resistance is encountered or the distance is equivalent to that from the ear to the nostril of the patient, indicating contact with the nasopharynx. Swab should reach depth equal to distance from nostrils to outer opening of the ear. Gently rub and roll the swab. Leave swab in place for several seconds to absorb secretions. Slowly remove swab while rotating it. Specimens can be collected from both sides using the same swab, but it is not necessary to collect specimens from both sides if the minitip is saturated with fluid from the first collection. If a deviated septum or blockage creates difficulty in obtaining the specimen from one nostril, use the same swab to obtain the specimen from the other nostril.
【HOW TO TEST】
Allow the test, specimen, buffer and/or controls to reach room temperature 15-30℃ (59-86℉) prior to testing.
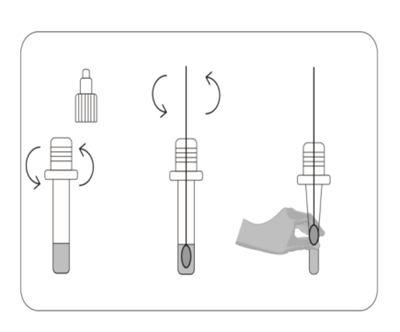
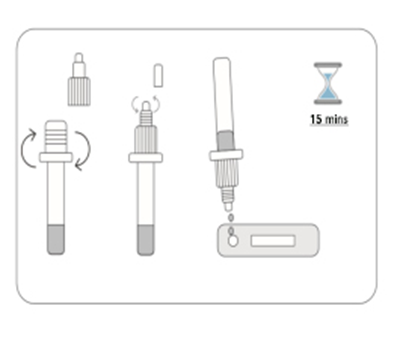
1.Unscrew the cap of the specimen extraction buffer. Use the Nasopharyngeal Swab to pick up fresh sample. Place the Nasopharyngeal Swab into the extraction buffer and shake and mix completely.
2.Take the test cassette from the packaging bag, place it on a table, cut off the protrusion of the collection tube, and add 2 drops of the sample into the sample hole vertically.
3. Read the result after 15 minutes. If left unread for 20 minutes or more the results are invalid and a repeat test is recommended.
【INTERPRETATION OF RESULTS】
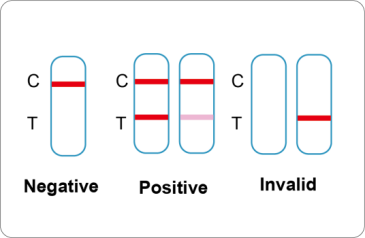
Positive: Two lines appear. One line should always appear in the control line region(C), and another one apparent colored line should appear in the test line region.
*NOTE: The intensity of the color in the test line regions may vary depending on the concentration of COVID-19 antibodies present in the specimen. Therefore, any shade of color in the test line region should be considered positive.
Negative: One colored line appears in the control region(C).No apparent colored line appear in the test line region.
Invalid: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test device. If the problem persists, discontinue using the test kit immediately and contact your local distributor.

1.jpg)
1-300x231.jpg)