Testsealabs Hot Sale!!! Thailand FDA Approval Most Popular GIGA Testsealabs Covid-19 Antigen Test Nasal $Saliva 2 in 1 Home Self-Test Kit
INTRODUCTION
The COVID-19 Antigen Test Cassette is a chromatographic immunoassay for the qualitative detection of SARS-CoV-2 nucleocapsid antigen in nasal swabs and saliva to aid in the diagnosis of SARS-CoV-2 virus infection.
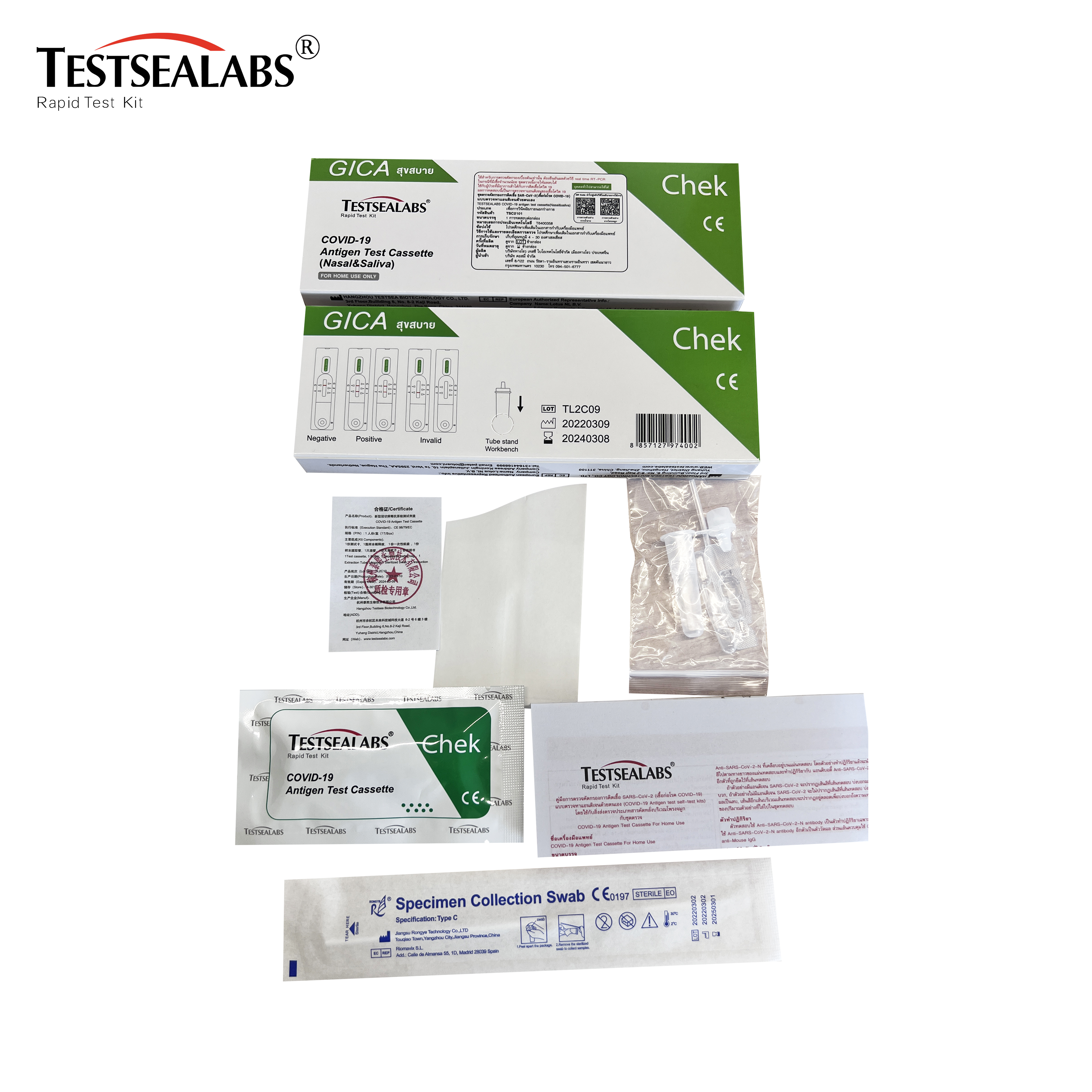
PRODUCT PICTURES
Notice!!! Legal one is in Thailand Package , if you are interesting in this one , you can contact Thailand Legal Distributor ----Suksabai Group Company!!!
Covid-19 antigen test 2 in 1 Suksabai Group Company order directly ship by air via, and the lot TL2C09,TL2C10,TL2C08 products they are selling are using our new diluent formulation, we call it A3 buffer , it can be more efficient to detection Omicron new variants BA.4, BA.5 & 2.75.

PRODUCT FEATURE
Fast and easy to self-test at anywhere
Easy to interpret the results using mobile application
Qualitatively detect the SARS-CoV-2 nucleocapsid protein
Use for nasal swab or saliva specimen
Fast results only in 10 minutes
Identify individual’s current infection status to COVID-19
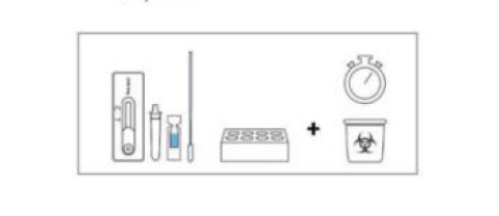
MATERIAL
Materials provided for Swab
Test device
Extraction buffer
Extraction tube
Sterile swab
Package insert
Workstation
Materials provided for Saliva
Test device
Extraction buffer
Extraction tube
Saliva Collection Bag
Dropper
Package insert
Workstation
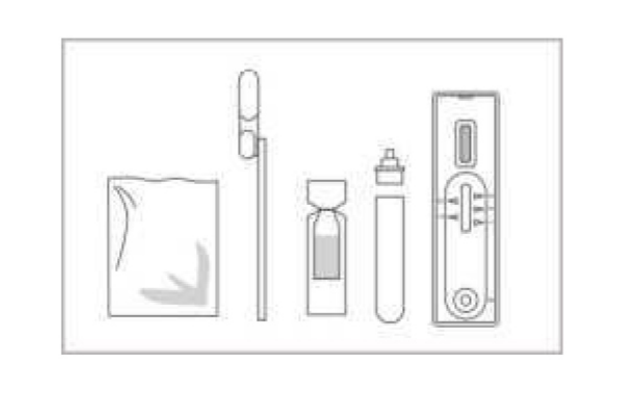
Materials required but not provided: Timer
SPECIMEN COLLECTION AND PREPARATION
The COVID-19 Antigen Test Cassette is designed for use with nasal swabs and saliva. For best results, a nasal swab is recommended.
Instructions for the nasopharyngea swab procedure
Insert the swab liberally trough the nose into the nasopharynx and swab the nasopharynx in 2-3 circular motions.
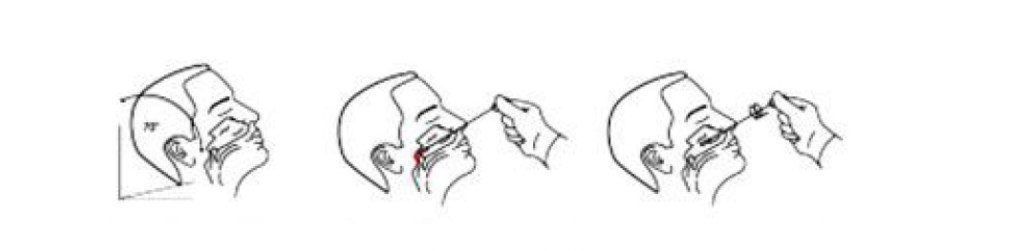
Instructions for the nasal swab procedure
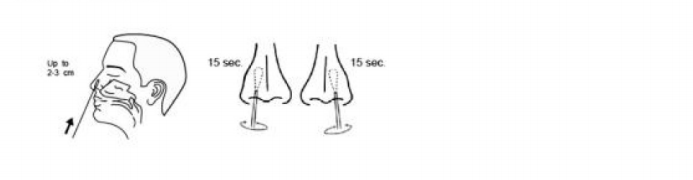
Insert the entire tip of the swab two to three centimetres into the left nostril. Rub the inside of the nostril in a circular motion for at least 15 seconds. Remove the swab and insert it into the right nostril. Swab the inside of the nostril in a circular motion for at least 15 seconds.
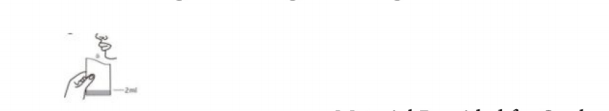
Instructions for Saliva Specimen procedure
Perform hand hygiene with soap and water/alcohol-based hand rub.Open the container. Make a “Kruuua” noise from the throat to clear the saliva from deep throat, then spit saliva(about 2ml) into the container. Avoid any saliva contamination of the outer surface of the container. Optimal timing of specimen collection: after getting up and before brushing teeth, eating or drinking.
DIRECTIONS FOR USE
Allow the test, sample and buffer to reach room temperature 15-30°C (59-86°F) before running.
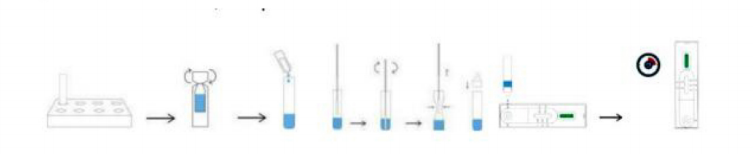
①Place the extraction tube in the tube stander.
②Unscrew the diluent bottle, open the extraction tube and pour all the extraction buffer into the extraction tube.
③Open the swab package then have nasal swab carried.
④Place the swab in the extraction tube. Rotate the swab for about 10 seconds while dripping the head against the inside of the tube to release the antigen in the swab.
⑤Remove the swab while pressing the head of the swab against the inside of the extraction tube to expel as much liquid as possible from the swab. Dispose of the swab according to the disposal regulations for biological waste.
⑥Screw the cap onto the extraction tube and make sure it is firmly in place.
⑦Place 3 drops of the sample vertically into the sample window of the test cassette. Read the result after 10-15 minutes. Read the result within 20 minutes. Otherwise, a repetition of the test is recommended.
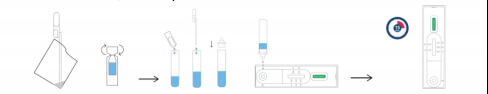
IRECTIONS FOR USE FOR SALIVA
1.Unscrew the diluent bottle,Unscrew the cap of the extraction tube, 2.Add all of the extraction buffer into the extraction tube
3.Transfer approximately 100uL of fresh saliva from container into the 4.Sample Extraction Tube and s hake and mix completely
5.Take the test cassette from the packaging bag, place it on a table, cut off the protrusion of the collection on tube, and add 3 drops of the sample into the sample hole vertically
6.Read the result after 15 minutes. If left unread for 20 minutes or more the results are invalid, and a rep eat test is recommended.
INTERPRETATION OF RESULTS
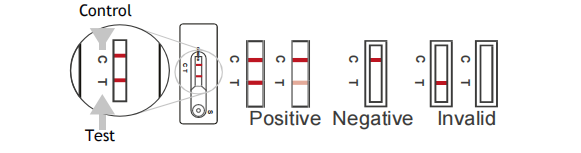
Positive: Two lines appear. One line should always appear in the control
line region(C), and another one apparent colored line should appear in
the test line region.
Negative: One colored line appear in the control region(C).No apparent
colored line appear in the test line region.
Invalid: Control line fail to appear. Insufficient specimen volume or
incorrect procedural techniques are the most likely reasons for control
line failure.