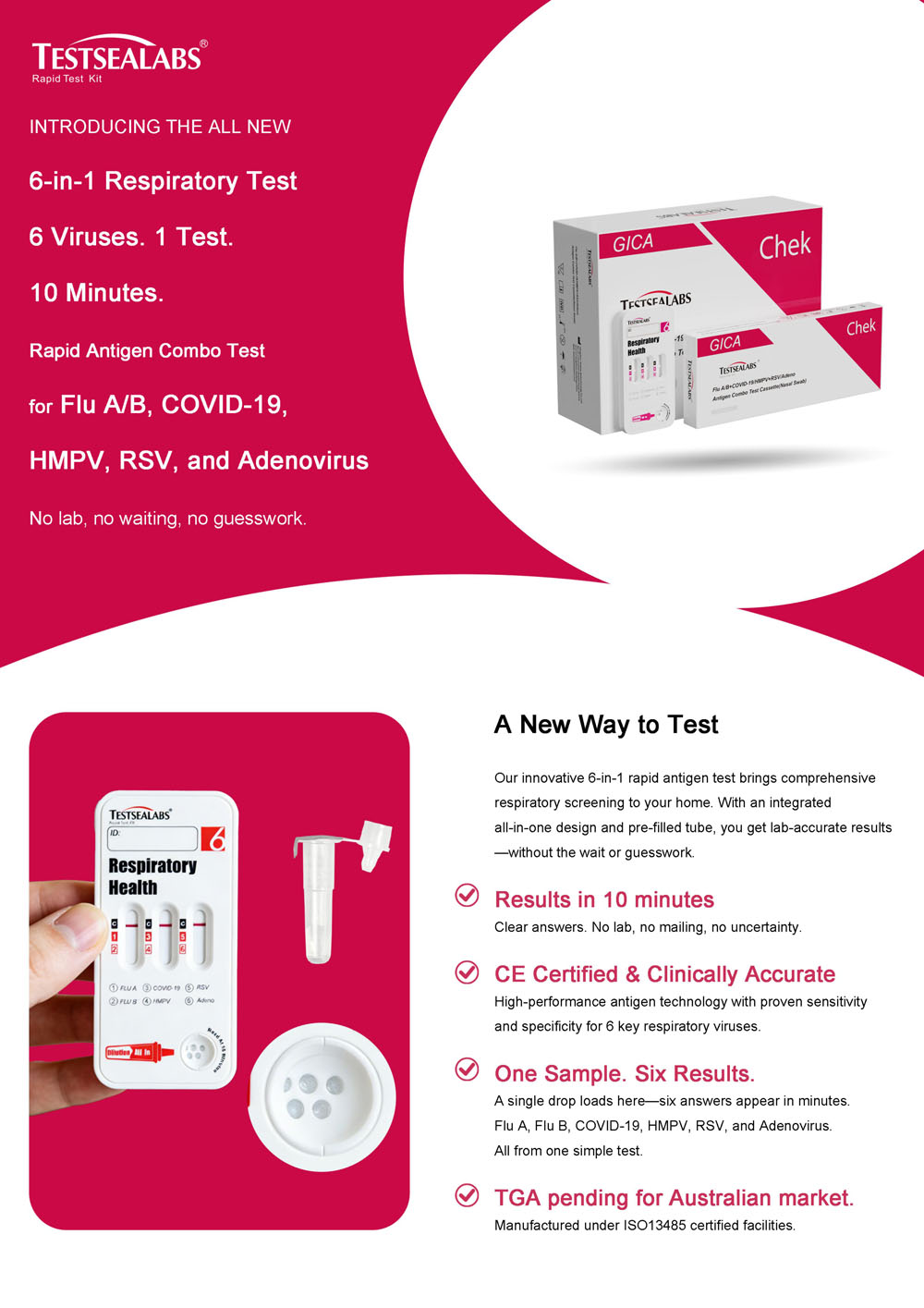
Hangzhou, China – April 22, 2026 – Hangzhou Testsea Biotechnology Co., Ltd., a leading global provider of in vitro diagnostic (IVD) solutions, today announced the official launch of its Flu A/B + COVID-19/HMPV + RSV/Adenovirus Antigen Combo Test Cassette, a revolutionary 3-in-1, 6-target rapid self-test designed for comprehensive at-home respiratory virus screening. The product has been officially approved by the Therapeutic Goods Administration (TGA) of Australia, with registration number ARTG 525693, marking a key milestone in the company’s global diagnostic product layout.
Product Overview
This nasal swab-based test simultaneously detects six major globally prevalent respiratory pathogens in a single test, covering the most common causes of acute respiratory infections:
- Influenza A (Flu A): Highly contagious, causes seasonal epidemics and global pandemics, with high risk of severe illness, pneumonia and complications in vulnerable populations.
- Influenza B (Flu B): Mainly circulates in human populations, triggers seasonal outbreaks and frequent cluster infections in families and schools.
- COVID-19 (SARS-CoV-2): Persistently mutating, causes a wide spectrum of respiratory illnesses, requiring rapid screening for timely isolation and public health protection.
- Human Metapneumovirus (HMPV): A common global cause of upper and lower respiratory infections, posing severe infection risks for infants, the elderly and immunocompromised individuals.
- Respiratory Syncytial Virus (RSV): The leading viral cause of severe lower respiratory tract infections in young children, also leading to severe symptoms in the elderly.
- Adenovirus: Causes febrile respiratory illness, pharyngitis, bronchitis and pneumonia, prone to outbreaks in closed settings such as schools and kindergartens.
Core Rationale for 6-Panel Combined Testing
These six pathogens account for the vast majority of acute respiratory infections worldwide, with highly overlapping clinical symptoms including fever, cough and runny nose, making them extremely difficult to distinguish by symptoms alone. While early symptoms are similar, corresponding treatment strategies vary significantly. Simultaneous multi-pathogen detection provides clear clinical guidance, and is highly valuable for family care decision-making, especially for the elderly and children.
Key Product Advantages
- Australian TGA Official Approval (ARTG 525693): The product has obtained authoritative international regulatory approval, ensuring reliable quality and safety for global household use.
- One Sample, Six Results, No Repeated Testing: Traditional home respiratory tests only target a single virus, forcing users to purchase multiple test kits or queue for hospital testing when symptomatic. This all-in-one test features an integrated cassette design and pre-assembled extraction tube, allowing users to complete one simple anterior nasal swab sample collection and read six test results simultaneously on one test card.
- Rapid 10-15 Minute Results: Delivers clear, reliable test results on-site within 10 to 15 minutes, with no laboratory equipment required.
- 6-in-1 Simultaneous Multi-Pathogen Detection: Unique combined panel design detects six viruses across three groups in one single test, realizing efficient one-stop screening.
- High Sensitivity and Specificity: Optimized limit of detection (LOD) for all viral targets, covering all major SARS-CoV-2 variants including Omicron, with stable and accurate clinical performance.
- User-Friendly At-Home Self-Testing: Non-invasive nasal swab collection, simplified operation steps, ideal for unsupervised self-testing in household settings.
- Clear and Intuitive Result Interpretation: The control line (C) validates test validity, and corresponding test lines indicate positive results for targeted pathogens, with easy-to-read results for all users.
Company Statement
“Many respiratory infections present nearly identical early symptoms, but require completely different treatment approaches,” said a spokesperson for Testsea Biotechnology. “We developed this 6-in-1 combo test to empower ordinary families to obtain clear, actionable diagnostic guidance quickly at home, without any laboratory equipment, supporting timely and appropriate health and care decisions.”
“As global demand for convenient, multi-target respiratory testing continues to rise, our innovative 3-in-1, 6-panel solution provides a reliable one-step screening tool for households, helping individuals and families better protect respiratory health efficiently.”
Clinical Performance
The combo test demonstrates excellent clinical accuracy, with outstanding sensitivity and specificity for all six targeted pathogens. It enables early identification of respiratory infections, timely isolation of infected individuals, and targeted clinical management, effectively supporting personal health monitoring and public health prevention efforts.
About Hangzhou Testsea Biotechnology Co., Ltd.
Headquartered in Hangzhou, China, Testsea Biotechnology is a professional IVD manufacturer dedicated to the R&D, production and sales of high-quality rapid antigen detection products. The company is committed to developing safe, accurate, user-friendly diagnostic solutions to support global public health and family health management.
Media Contact
Hangzhou Testsea Biotechnology Co., Ltd.
No.13-2 Guanshan Road, Yuhang District, Hangzhou, Zhejiang 311115, China
Website: www.testsealabs.com
Disclaimer
This product is for in vitro diagnostic (IVD) use only. Test results are for preliminary screening reference only and do not constitute a definitive medical diagnosis. For formal medical diagnosis, treatment and health guidance, please consult a qualified healthcare professional.
Post time: Apr-23-2026