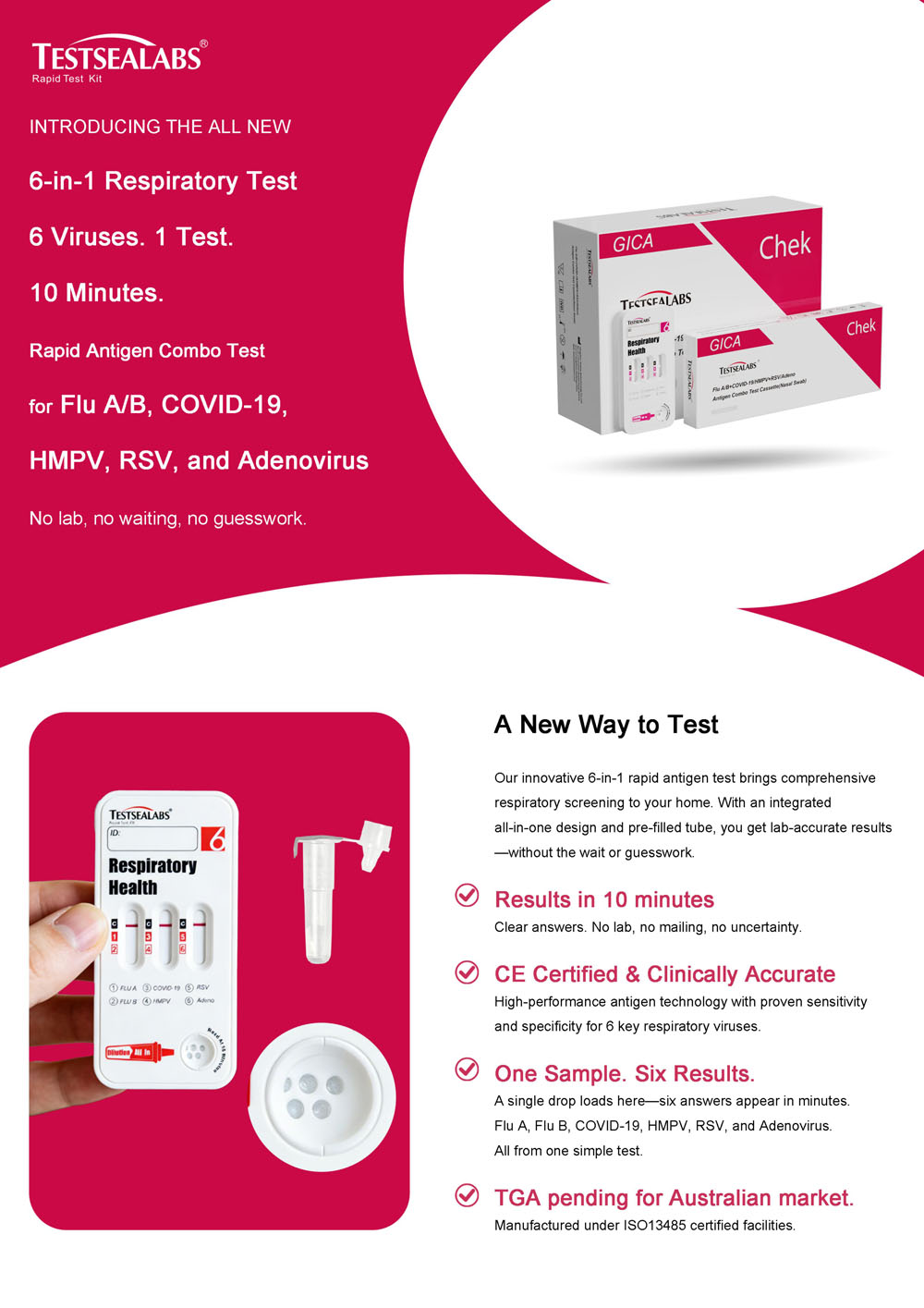
Australia TGA Flu A/B + COVID-19/HMPV+RSV/Adeno Antigen Combo Test Cassette (Nasal Swab)
Product Specification
| Principle | Chromatographic Immunoassay | Specimen | Nasal Swab |
| Reading Time | 10 minutes | QMS Certification | ISO 13485 and MDSAP |
| Storage Temperature | 4-30°C | Shelf life | 2 years |
| Format | Cassette | Specification | 1T/5T/25T |
Intended Use
A rapid test for the qualitative detection of influenza A virus,influenza Bvirus,COVID-19, human metapneumovirus, respiratory syncytial virusand adenovirus antigen in nasal swab specimens.
Summary
Various respiratory conditions, including chronic and infectious conditions, can hinder your ability to breathe. Diseases that affect the lungs include cancer, pneumonia and other inflammatory diseases. Chronic lung diseases fall into one of two main classes: obstructive or restrictive. People with weakened lungs and immune systems are more vulnerable to infectious respiratory conditions. Some individuals have multiple lung conditions. An antigen is generally detectable in upper respiratory specimens during the acute phase of infection. Positive results indicate the presence of viral antigen, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status. Positive results do not rule out other bacterial/viral infection. Negative results should be treated as presumptive and confirmed with a molecular assay, if necessary for patient management. Negative results should be considered in the context of a patient’s recent exposures, history and the presence of clinical signs and symptoms consistent with influenza A+B, COVID-19, HMPV, RSV and Adeno. The Flu A/B + COVID-19/HMPV + RSV/Adeno Antigen Combo Test Cassette is intended for use by trained clinical laboratory personnel.